Abstract
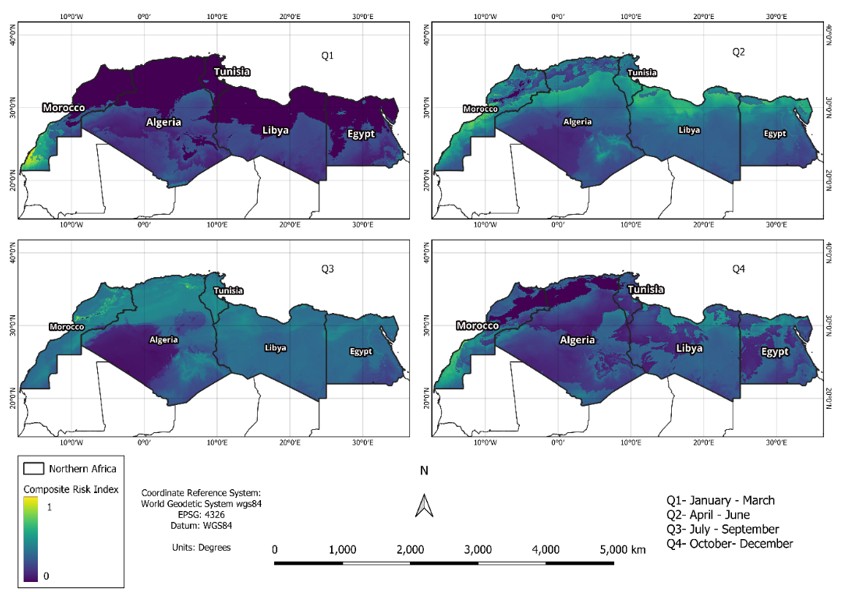
In vector-borne disease modelling, combining temperature-sensitive vectorial capacity with population growth dynamics often leads to complex frameworks that are difficult to interpret and operationalize. We propose a biologically grounded yet parsimonious approach that integrates the dominant eigenvalue of a temperature-driven next-generation matrix (λmax) with a relative vectorial capacity (VC*) function to estimate the seasonal climate-driven transmission risk (Q1; January–March, Q2; April–June, Q3; July–September, Q3; October–December) of zoonotic cutaneous leishmaniasis mediated by Phlebotomus papatasi across North Africa. Across multiple composite formulations, robust discrimination and spatial reliability were concentrated almost exclusively in Q3, during which additive model weighting 90% VC* and 10% λmax achieved the highest overall performance (mean AUC-PR = 0.67 ± 0.09; TSS = 0.54; Boyce = 0.85). Outside Q3, predictive skill deteriorated sharply, indicating that seasonal ecological constraints impose a fundamental limit on model performance that cannot be offset by aggregation strategy alone. Uncertainty-aware mid-century projections under SSP2-4.5 and SSP5-8.5, generated via Monte Carlo propagation of temperature-dependent entomological traits, reveal robust spatial risk patterns but strong seasonal heterogeneity in uncertainty. Paired analyses at 581 endemic sites show highly significant seasonal shifts in climatic suitability in Q1–Q3 (Wilcoxon p < 0.001), with consistent increases in Q1, strong decreases in Q2–Q3, and weaker, scenario-dependent changes in Q4. Overlaying climatic suitability with healthcare accessibility further reveals persistent spatial mismatches between ecological risk and health system capacity. Together, these results highlight the value of interpretable, seasonally explicit, and uncertainty-aware frameworks for climate-informed disease risk assessment in data-limited settings.
References
Agboka, K.M., Diallo, S., Hassaballa, I.B., Meltus, Q.J., Ahmed, K., Tanga, C.M., Landmann, T., & Abdel-Rahman, E.M. (2026) Temperature-and host-driven model of Phlebotomus papatasi outbreak potential under climate change in Sudan. Ecological Modelling 511, 111386. https://doi.org/10.1016/j.ecolmodel.2025.111386.
Agboka, K.M., Ndjomatchoua, F.T., Guimapi, R.A., Rossini, L., Azrag, A.G.A., Meltus, Q.J., Landmann, T., Ekesi, S., & Abdel-Rahman, E.M. (2025a) Dual Perspectives From Mechanistic and Correlative Approaches in Mapping the Distribution of the Lesser Mealworm Alphitobius diaperinus. Physiol Plant. 177(6), e70659. doi: 10.1111/ppl.70659. PMID: 41320850.
Agboka, K. M., Ndjomatchoua, F. T., Rossini, L., Guimapi, R. A., & Abdel-Rahman, E. M. (2025b). When thermal risk indices work and when they don't: A case study of two maize insect pests. MethodsX, 15, 103537. https://doi.org/10.1016/j.mex.2025.103537.
Ahmed, A., Ali, Y., & Mohamed, N. S. (2020). Arboviral diseases: the emergence of a major yet ignored public health threat in Africa. The Lancet. Planetary health, 4(12), e555. https://doi.org/10.1016/S2542-5196(20)30269-2.
Aoun, K., & Bouratbine, A. (2014). Cutaneous leishmaniasis in North Africa: a review. Parasite (Paris, France), 21, 14. https://doi.org/10.1051/parasite/2014014.
Babaousmail, H., Hou, R., Ayugi, B., Ojara, M., Ngoma, H., Karim, R., Rajasekar, A., & Ongoma, V. (2021). Evaluation of the Performance of CMIP6 Models in Reproducing Rainfall Patterns over North Africa. Atmosphere, 12(4), 475. https://doi.org/10.3390/atmos12040475.
Bates P. A. (2007). Transmission of Leishmania metacyclic promastigotes by phlebotomine sand flies. International journal for parasitology, 37(10), 1097–1106. https://doi.org/10.1016/j.ijpara.2007.04.003.
Benkova, I., & Volf, P. (2007). Effect of temperature on metabolism of Phlebotomus papatasi (Diptera: Psychodidae). Journal of medical entomology, 44(1), 150–154. https://doi.org/10.1603/0022-2585(2007)44[150:eotomo]2.0.co;2.
Berry, I., & Berrang-Ford, L. (2016). Leishmaniasis, conflict, and political terror: A spatio-temporal analysis. Social Science & Medicine, 167, 140-149.
Bounoua, L., Kahime, K., Houti, L., Blakey, T., Ebi, K. L., Zhang, P., Imhoff, M. L., Thome, K. J., Dudek, C., Sahabi, S. A., Messouli, M., Makhlouf, B., El Laamrani, A., & Boumezzough, A. (2013). Linking climate to incidence of zoonotic cutaneous leishmaniasis (L. major) in pre-Saharan North Africa. International Journal of Environmental Research and Public Health, 10(8), 3172–3191. https://doi.org/10.3390/ijerph10083172.
Bozorg-Omid, F., Youssefi, F., Hassanpour, G., Rahimi Foroushani, A., Rahimi, M., Shirzadi, M. R., Jafari, R., & Hanafi-Bojd, A. A. (2025). Predicting the Effect of Temperature Changes on Phlebotomus papatasi Activity, as the Main Vector of Zoonotic Cutaneous Leishmaniasis in Iran. Transboundary and Emerging Diseases, 2025, 9518371. https://doi.org/10.1155/tbed/9518371.
Brady, O. J., Godfray, H. C., Tatem, A. J., Gething, P. W., Cohen, J. M., McKenzie, F. E., Perkins, T. A., Reiner, R. C., Jr, Tusting, L. S., Sinka, M. E., Moyes, C. L., Eckhoff, P. A., Scott, T. W., Lindsay, S. W., Hay, S. I., & Smith, D. L. (2016). Vectorial capacity and vector control: reconsidering sensitivity to parameters for malaria elimination. Transactions of the Royal Society of Tropical Medicine and Hygiene, 110(2), 107–117. https://doi.org/10.1093/trstmh/trv113.
Buckingham-Jeffery, E., Hill, E. M., Datta, S., Dilger, E., & Courtenay, O. (2019). Spatio-temporal modelling of Leishmania infantum infection among domestic dogs: a simulation study and sensitivity analysis applied to rural Brazil. Parasites & Vectors, 12(1), 215. https://doi.org/10.1186/s13071-019-3430-y.
Carlson, C. J., Albery, G. F., Merow, C., Trisos, C. H., Zipfel, C. M., Eskew, E. A., Olival, K. J., Ross, N., & Bansal, S. (2022). Climate change increases cross-species viral transmission risk. Nature, 607(7919), 555–562. https://doi.org/10.1038/s41586-022-04788-w.
Ceccato, P., Vancutsem, C., Klaver, R., Rowland, J., & Connor, S. J. (2012). A vectorial capacity product to monitor changing malaria transmission potential in epidemic regions of Africa. Journal of Tropical Medicine, 2012, 595948. https://doi.org/10.1155/2012/595948.
Chamberlain, S. A., & Boettiger, C. (2017). R Python, and Ruby clients for GBIF species occurrence data. PeerJ Preprints.
Charrahy, Z., Yaghoobi-Ershadi, M. R., Shirzadi, M. R., Akhavan, A. A., Rassi, Y., Hosseini, S. Z., Webb, N. J., Haque, U., Bozorg Omid, F., & Hanafi-Bojd, A. A. (2022). Climate change and its effect on the vulnerability to zoonotic cutaneous leishmaniasis in Iran. Transboundary and Emerging Diseases, 69(3), 1506–1520. https://doi.org/10.1111/tbed.14115.
Chelbi, I., & Zhioua, E. (2007). Biology of Phlebotomus papatasi (Diptera: Psychodidae) in the laboratory. Journal of Medical Entomology, 44(4), 597–600. https://doi.org/10.1603/0022-2585(2007)44[597:boppdp]2.0.co;2.
Colacicco-Mayhugh, M. G., Masuoka, P. M., & Grieco, J. P. (2010). Ecological niche model of Phlebotomus alexandri and P. papatasi (Diptera: Psychodidae) in the Middle East. International Journal of Health Geographics, 9, 2. https://doi.org/10.1186/1476-072X-9-2.
Dennington, N. L., Grossman, M. K., Ware-Gilmore, F., Teeple, J. L., Johnson, L. R., Shocket, M. S., McGraw, E. A., & Thomas, M. B. (2024). Phenotypic adaptation to temperature in the mosquito vector, Aedes aegypti. Global Change Biology, 30(1), e17041. https://doi.org/10.1111/gcb.17041.
Diekmann, O., Heesterbeek, J. A., & Metz, J. A. (1990). On the definition and the computation of the basic reproduction ratio R0 in models for infectious diseases in heterogeneous populations. Journal of Mathematical Biology, 28(4), 365–382. https://doi.org/10.1007/BF00178324.
Du, R., Hotez, P. J., Al-Salem, W. S., & Acosta-Serrano, A. (2016). Old World Cutaneous Leishmaniasis and Refugee Crises in the Middle East and North Africa. PLoS Neglected Tropical Diseases, 10(5), e0004545. https://doi.org/10.1371/journal.pntd.0004545.
El Omari, H., Chahlaoui, A., Talbi, F. Z., Mouhdi, K. E., & El Ouali Lalami, A. (2020). Impact of Climatic Factors on the Seasonal Fluctuation of Leishmaniasis Vectors in Central Morocco (Meknes Prefecture). The Canadian Journal of Infectious Diseases & Medical Microbiology, 2020, 6098149. https://doi.org/10.1155/2020/6098149.
Fick, S.E. and Hijmans, R.J. (2017), WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol, 37: 4302-4315. https://doi.org/10.1002/joc.5086.
Garrett-Jones, C. (1964). Prognosis for interruption of malaria transmission through assessment of the mosquito's vectorial capacity. Nature, 204, 1173–1175. https://doi.org/10.1038/2041173a0.
Gherbi, R., Bounechada, M., Latrofa, M. S., Annoscia, G., Tarallo, V. D., Dantas-Torres, F., & Otranto, D. (2020). Phlebotomine sand flies and Leishmania species in a focus of cutaneous leishmaniasis in Algeria. PLoS Neglected Tropical Diseases, 14(2), e0008024. https://doi.org/10.1371/journal.pntd.0008024.
Halimi, M., Cheghabaleki, Z. Z., Modrek, M. J., & Delavari, M. (2016). Temporal Dynamics of Phlebotomine Sand Flies Population in Response to Ambient Temperature Variation, Bam, Kerman Province of Iran. Annals of Global Health, 82(5), 824–831. https://doi.org/10.1016/j.aogh.2016.07.009.
Hanafi-Bojd, A. A., Yaghoobi-Ershadi, M. R., Haghdoost, A. A., Akhavan, A. A., Rassi, Y., Karimi, A., & Charrahy, Z. (2015). Modeling the Distribution of Cutaneous Leishmaniasis Vectors (Psychodidae: Phlebotominae) in Iran: A Potential Transmission in Disease Prone Areas. Journal of Medical Entomology, 52(4), 557–565. https://doi.org/10.1093/jme/tjv058.
Hand, D. J. (2007). Principles of data mining. Drug Safety, 30(7), 621–622. https://doi.org/10.2165/00002018-200730070-00010.
Hengl, T., Heuvelink, G. B. M., Kempen, B., Leenaars, J. G. B., Walsh, M. G., Shepherd, K. D., Sila, A., MacMillan, R. A., Mendes de Jesus, J., Tamene, L., & Tondoh, J. E. (2015) Mapping Soil Properties of Africa at 250 m Resolution: Random Forests Significantly Improve Current Predictions. PLOS ONE 10(6), e0125814. https://doi.org/10.1371/journal.pone.0125814.
Hotez, P. J., Savioli, L., & Fenwick, A. (2012). Neglected tropical diseases of the Middle East and North Africa: review of their prevalence, distribution, and opportunities for control. PLoS Neglected Tropical Diseases, 6(2), e1475. https://doi.org/10.1371/journal.pntd.0001475.
Hou, H., Zhou, B.-B., Pei, F., Hu, G., Su, Z., Zeng, Y., et al. (2022). Future land use/land cover change has nontrivial and potentially dominant impact on global gross primary productivity. Earth's Future, 10, e2021EF002628. https://doi.org/10.1029/2021EF002628.
Hua, R., Su, Q., Fan, J., Wang, L., Xu, L., Hui, Y., Huang, M., Du, B., Tian, Y., Zhao, Y., & Manduriwa (2025). Temporal and Spatial Dynamics of Rodent Species Habitats in the Ordos Desert Steppe, China. Animals, 15(5), 721. https://doi.org/10.3390/ani15050721.
Huete, A., Justice, C., & Van Leeuwen, W. (1999). MODIS vegetation index (MOD13). Algorithm theoretical basis document, 3(213), 295-309.
Karmaoui, A., Sereno, D., El Jaafari, S., & Hajji, L. (2022a). Seasonal patterns of zoonotic cutaneous leishmaniasis caused by L. major and transmitted by Phlebotomus papatasi in the North Africa region, a systematic review and a meta-analysis. Microorganisms, 10(12), 2391.
Karmaoui, A., Ben Salem, A., Sereno, D., El Jaafari, S., & Hajji, L. (2022b). Geographic distribution of Meriones shawi,Psammomys obesus, and Phlebotomus papatasi the main reservoirs and principal vector of zoonotic cutaneous leishmaniasis in the Middle East and North Africa. Parasite Epidemiology and Control, 17, e00247. https://doi.org/10.1016/j.parepi.2022.e00247.
Kasap, O. E., & Alten, B. (2005). Laboratory estimation of degree-day developmental requirements of Phlebotomus papatasi (Diptera: Psychodidae). Journal of Vector Ecology, 30(2), 328–333.
Lagerwall, G., Kiker, G., Muñoz-Carpena, R., & Wang, N. (2014). Global uncertainty and sensitivity analysis of a spatially distributed ecological model. Ecological Modelling, 275, 22-30. https://doi.org/10.1016/j.ecolmodel.2013.12.010.
Lawyer, P. G., Githure, J. I., Anjili, C. O., Olobo, J. O., Koech, D. K., & Reid, G. D. (1990). Experimental transmission of Leishmania major to vervet monkeys (Cercopithecus aethiops) by bites of Phlebotomus duboscqi (Diptera: Psychodidae). Transactions of the Royal Society of Tropical Medicine and Hygiene, 84(2), 229–232. https://doi.org/10.1016/0035-9203(90)90266-h.
Macdonald. G, M. G. (1957). The epidemiology and control of malaria.
Miller, E., Warburg, A., Novikov, I., Hailu, A., Volf, P., Seblova, V., & Huppert, A. (2014). Quantifying the contribution of hosts with different parasite concentrations to the transmission of visceral leishmaniasis in Ethiopia. PLoS Neglected Tropical Diseases, 8(10), e3288. https://doi.org/10.1371/journal.pntd.0003288.
Mordecai, E. A., Caldwell, J. M., Grossman, M. K., Lippi, C. A., Johnson, L. R., Neira, M., Rohr, J. R., Ryan, S. J., Savage, V., Shocket, M. S., Sippy, R., Stewart Ibarra, A. M., Thomas, M. B., & Villena, O. (2019). Thermal biology of mosquito-borne disease. Ecology Letters, 22(10), 1690–1708. https://doi.org/10.1111/ele.13335.
Ndjomatchoua, F. T., Guimapi, R. A. Y., Rossini, L., Djouda, B. S., & Pedro, S. A. (2024). A generalized risk assessment index for forecasting insect population under the effect of temperature. Journal of Thermal Biology, 122, 103886. https://doi.org/10.1016/j.jtherbio.2024.103886.
Parham, P. E., & Michael, E. (2010). Modelling climate change and malaria transmission. Advances in Experimental Medicine and Biology, 673, 184–199. https://doi.org/10.1007/978-1-4419-6064-1_13.
Parkash V, Ashwin H, Sadlova J, Vojtkova B, Jones G, Martin N, Greensted E, Allgar V, Kamhawi S, Valenzuela JG, Layton AM, Jaffe CL, Volf P, Kaye PM, Lacey CJN. A clinical study to optimise a sand fly biting protocol for use in a controlled human infection model of cutaneous leishmaniasis (the FLYBITE study). Wellcome Open Res. 2021 Jun 30;6:168. doi: 10.12688/wellcomeopenres.16870.1. PMID: 34693027; PMCID: PMC8506224.
Quiñonez-Díaz, L., Mancilla-Ramírez, J., Avila-García, M., Ortiz-Avalos, J., Berron, A., González, S., Paredes, Y., & Galindo-Sevilla, N. (2012). Effect of ambient temperature on the clinical manifestations of experimental diffuse cutaneous leishmaniasis in a rodent model. Vector borne and zoonotic diseases (Larchmont, N.Y.), 12(10), 851–860. https://doi.org/10.1089/vbz.2011.0844.
R Core Team. (2025). R Foundation for Statistical Computing, Vienna, Austria.
Rigosi, A., & Rueda, F. J. (2012). Propagation of uncertainty in ecological models of reservoirs: From physical to population dynamic predictions. Ecological Modelling, 247, 199-209.https://doi.org/10.1016/j.ecolmodel.2012.08.022.
Rizzoli, A., Tagliapietra, V., Cagnacci, F., Marini, G., Arnoldi, D., Rosso, F., & Rosà, R. (2019). Parasites and wildlife in a changing world: The vector-host- pathogen interaction as a learning case. International Journal for Parasitology. Parasites and wildlife, 9, 394–401. https://doi.org/10.1016/j.ijppaw.2019.05.011.
Rossini, L., & Bruzzone, O. A. (2025). A novel PDE model to describe terrestrial arthropods considering physiological age, reproduction rate, and body mass. Acta IMEKO, 14(1), 1-11. https://doi.org/10.21014/actaimeko.v14i1.1873.
Rossini, L., Bruzzone, O. A., Contarini, M., Bufacchi, L., & Speranza, S. (2022). A Physiologically Based ODE Model for an Old Pest: Modeling Life Cycle and Population Dynamics of Bactrocera oleae (Rossi). Agronomy, 12(10), 2298. https://doi.org/10.3390/agronomy12102298.
Rossini, L., Contarini, M., Bono Rosselló, N., Garone, E., & Speranza, S. (2022, September). Prediction of infestations by true bugs in hazelnut orchards: feasibility and preliminary approaches in the case of Halyomorpha halys. In X International Congress on Hazelnut 1379 (pp. 463-472). https://doi.org/10.17660/ActaHortic.2023.1379.66.
Rossini, L., Contarini, M. & Speranza, S. A novel version of the Von Foerster equation to describe poikilothermic organisms including physiological age and reproduction rate. Ricerche mat 70, 489–503 (2021). https://doi.org/10.1007/s11587-020-00489-6.
Rossini, L., Contarini, M., Speranza, S., Mermer, S., Walton, V., Francis, F., & Garone, E. (2024). Life tables in entomology: A discussion on tables' parameters and the importance of raw data. PloS One, 19(3), e0299598. https://doi.org/10.1371/journal.pone.0299598.
Rossini, L., Lots, A., Noël, G., Segers, A., Mermer, S., Contarini, M., Speranza, S., Walton, V., Francis, F., & Garone, E. (2025). Life tables data collection in entomology: an overview on the differential and the integral representation and proposal for a standard electronic file. Insect Science, 10.1111/1744-7917.70040. https://doi.org/10.1111/1744-7917.70040.
Rossini, L., Rosselló, N. B., Contarini, M., Speranza, S., & Garone, E. (2022). Modelling ectotherms’ populations considering physiological age structure and spatial motion: A novel approach. Ecological Informatics, 70, 101703. https://doi.org/10.1016/j.ecoinf.2022.101703.
Rossini, L., Severini, M., Contarini, M., & Speranza, S. (2019). A novel modelling approach to describe an insect life cycle vis-à-vis plant protection: description and application in the case study of Tuta absoluta. Ecological Modelling, 409, 108778. https://doi.org/10.1016/j.ecolmodel.2019.108778.
Rounsevell, M. D., Arneth, A., Brown, C., Cheung, W. W., Gimenez, O., Holman, I., ... & Shin, Y. J. (2021). Identifying uncertainties in scenarios and models of socio-ecological systems in support of decision-making. One Earth, 4(7), 967-985. https://doi.org/10.1016/j.oneear.2021.06.003.
Suh, E., Grossman, M. K., Waite, J. L., Dennington, N. L., Sherrard-Smith, E., Churcher, T. S., & Thomas, M. B. (2020). The influence of feeding behaviour and temperature on the capacity of mosquitoes to transmit malaria. Nature Ecology & Evolution, 4(7), 940–951. https://doi.org/10.1038/s41559-020-1182-x.
Talisuna, A. O., Okiro, E. A., Yahaya, A. A., Stephen, M., Bonkoungou, B., Musa, E. O., Minkoulou, E. M., Okeibunor, J., Impouma, B., Djingarey, H. M., Yao, N. K. M., Oka, S., Yoti, Z., & Fall, I. S. (2020). Spatial and temporal distribution of infectious disease epidemics, disasters and other potential public health emergencies in the World Health Organisation Africa region, 2016-2018. Globalization and Health, 16(1), 9. https://doi.org/10.1186/s12992-019-0540-4.
Terradas, G., Manzano-Alvarez, J., Vanalli, C., Werling, K., Cattadori, I. M., & Rasgon, J. L. (2024). Temperature affects viral kinetics and vectorial capacity of Aedes aegypti mosquitoes co-infected with Mayaro and Dengue viruses. Parasites & Vectors, 17(1), 73. https://doi.org/10.1186/s13071-023-06109-0.
Thompson, R. N., Gilligan, C. A., & Cunniffe, N. J. (2020). Will an outbreak exceed available resources for control? Estimating the risk from invading pathogens using practical definitions of a severe epidemic. Journal of the Royal Society, Interface, 17(172), 20200690. https://doi.org/10.1098/rsif.2020.0690.
van den Driessche, P., & Watmough, J. (2002). Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Mathematical Biosciences, 180, 29–48. https://doi.org/10.1016/s0025-5564(02)00108-6.
Wilson, A. J., Morgan, E. R., Booth, M., Norman, R., Perkins, S. E., Hauffe, H. C., ... & Fenton, A. (2017). What is a vector?. Philosophical Transactions of the Royal Society B: Biological Sciences, 372(1719). https://doi.org/10.1098/rstb.2016.0085.
Woolson, R.F. (2008). Wilcoxon Signed-Rank Test. In Wiley Encyclopedia of Clinical Trials (eds R.B. D'Agostino, L. Sullivan and J. Massaro). https://doi.org/10.1002/9780471462422.eoct979.
World Health Organization. Leishmaniasis Fact Sheet. Geneva: World Health Organization; (2023). [cited 2025 Jun 20]. Available from: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis.
Yaghoobi-Ershadi, M. R., Shirani-Bidabadi, L., Hanafi-Bojd, A. A., Akhavan, A. A., & Zeraati, H. (2007). Colonization and Biology of Phlebotomus papatasi, the Main Vector of Cutaneous Leishmaniasis due to Leishmania major. Iran J Public Health, 36(3), 21–26. https://ijph.tums.ac.ir/index.php/ijph/article/view/1735.
Zomer, R.J., Xu, J. & Trabucco, A. (2022) Version 3 of the Global Aridity Index and Potential Evapotranspiration Database. Scientific Data 9, 409. https://doi.org/10.1038/s41597-022-01493-1.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright (c) 2026 Komi M. Agboka, Souleymane Diallo, Steve B.S. Baleba, Iman B. Hassaballa, Quinto J. Meltus, Allan M. Ngángá, Harriet W. Wangu, Khalid Ahmed, Chrysantus M. Tanga, Tobias Landmann, and Elfatih M. Abdel-Rahman